Olympus Corporation acquired Medi-Tate Ltd., an Israeli medical device company.
The amount of the deal was not disclosed.
Through the acquisition, Olympus expands its business line in offering in-office treatment for benign prostatic hyperplasia (BPH) treatment and solidifies its position as a leader in the field of urological devices.
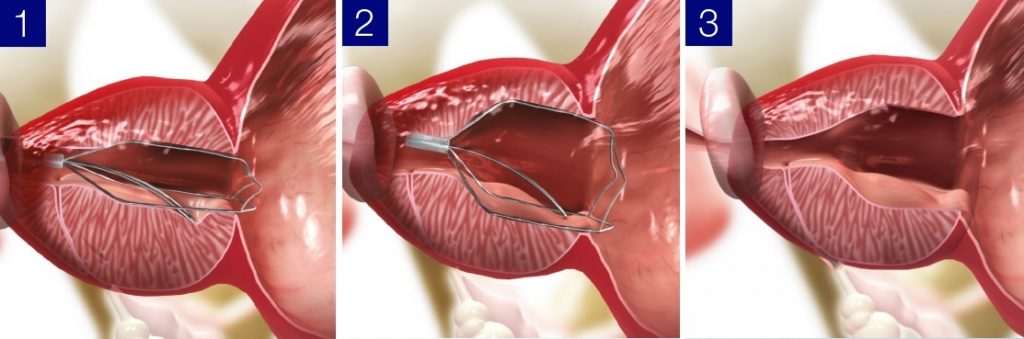
Led by CEO Ido Kilemnik, Medi-Tate is a medical device manufacturer engaged in the research and development, production, and sale of devices for the minimally invasive treatment of BPH. The flagship iTind treatment for BPH currently has the EU CE Mark and is approved for sale in the European Union, UK, Israel, Australia, Brazil, and the FDA cleared for use in the United States.
With its initial Medi-Tate investment, Olympus has had the right of distribution since November 2018.
Olympus provides devices for BPH treatment such as resectoscopes and a wide variety of electrodes. By adding the non-ablative BPH treatment device iTind, which allows patients to maintain their sexual function, to its portfolio, Olympus can provide urologists more treatment options according to the symptoms and needs of their patients, ranging from in-offices day treatment to surgical therapies in hospitals.
FinSMEs
28/05/2021